 常见物质溶解度.docx
常见物质溶解度.docx
- 文档编号:6163309
- 上传时间:2023-01-04
- 格式:DOCX
- 页数:17
- 大小:21.22KB
常见物质溶解度.docx
《常见物质溶解度.docx》由会员分享,可在线阅读,更多相关《常见物质溶解度.docx(17页珍藏版)》请在冰豆网上搜索。
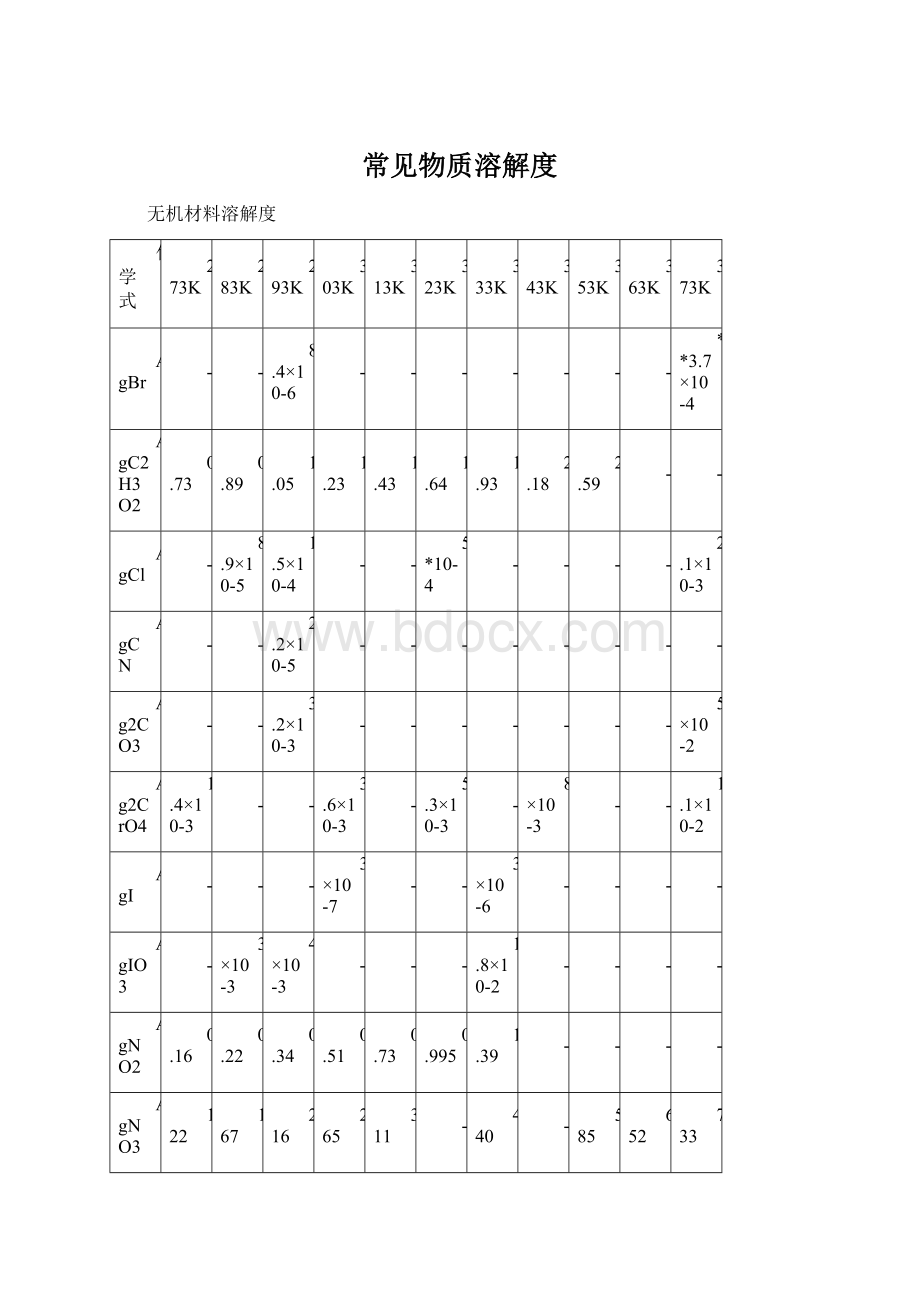
常见物质溶解度
无机材料溶解度
化学式
273K
283K
293K
303K
313K
323K
333K
343K
353K
363K
373K
AgBr
-
-
8.4×10-6
-
-
-
-
-
-
-
**3.7×10-4
AgC2H3O2
0.73
0.89
1.05
1.23
1.43
1.64
1.93
2.18
2.59
-
-
AgCl
-
8.9×10-5
1.5×10-4
-
-
5*10-4
-
-
-
-
2.1×10-3
AgCN
-
-
2.2×10-5
-
-
-
-
-
-
-
-
Ag2CO3
-
-
3.2×10-3
-
-
-
-
-
-
-
5×10-2
Ag2CrO4
1.4×10-3
-
-
3.6×10-3
-
5.3×10-3
-
8×10-3
-
-
1.1×10-2
AgI
-
-
-
3×10-7
-
-
3×10-6
-
-
-
-
AgIO3
-
3×10-3
4×10-3
-
-
-
1.8×10-2
-
-
-
-
AgNO2
0.16
0.22
0.34
0.51
0.73
0.995
1.39
-
-
-
-
AgNO3
122
167
216
265
311
-
440
-
585
652
733
Ag2SO4
0.57
0.7
0.8
0.89
0.98
1.08
1.15
1.22
1.3
1.36
1.41
AlCl3
43.9
44.9
45.8
46.6
47.3
-
48.1
-
48.6
-
49
AlF3
0.56
0.56
0.67
0.78
0.91
-
1.1
-
1.32
-
1.72
Al(NO3)3
60
66.7
73.9
81.8
88.7
-
106
-
132
153
160
Al2(SO4)3
31.2
33.5
36.4
40.4
45.8
52.2
59.2
66.1
73
80.8
89
As2O5
59.5
62.1
65.8
69.8
71.2
-
73
-
75.1
-
76.7
As2S5
-
-
5.17×10-5(291)
-
-
-
-
-
-
-
-
B2O3
1.1
1.5
2.2
-
4
-
6.2
-
9.5
-
15.7
BaCl2·2H2O
31.2
33.5
35.8
38.1
40.8
43.6
46.2
49.4
52.5
55.8
59.4
BaCO3
-
1.6×10-3(281)
2.2×10-3(291)
2.4×10-3(297.2)
-
-
-
-
-
-
6.5×10-3
BaC2O4
-
-
9.3×10-3(291)
-
-
-
-
-
-
-
2.28×10-2
BaCrO4
2.0×10-4
2.8×10-4
3.7×10-4
4.6×10-4
-
-
-
-
-
-
-
Ba(NO3)2
4.95
6.67
9.02
11.48
14.1
17.1
20.4
-
27.2
-
34.4
Ba(OH)2
1.67
2.48
3.89
5.59
8.22
13.12
20.94
-
101.4
-
-
BaSO4
1.15×10-4
2.0×10-4
2.4×10-4
2.85×10-4
-
3.36×10-4
-
-
-
-
4.13×10-4
BeSO4
37
37.6
39.1
41.4
45.8
-
53.1
-
67.2
-
82.8
Br2
4.22
3.4
3.2
3.13
-
-
-
-
-
-
-
Bi2S3
-
-
1.8×10-5(291)
-
-
-
-
-
-
-
-
CaBr2·6H2O
125
132
143
185(307)
213
-
278
-
295
-
312(378)
Ca(H2C3O2)2·2H2O
37.4
36
34.7
33.8
33.2
-
32.7
-
33.5
-
-
CaCl2·6H2O
59.5
64.7
74.5
100
128
-
137
-
147
154
159
CaC2O4
-
6.7×10-4(286)
6.8×10-4(298)
-
-
9.5×10-4
-
-
-
14×10-4(368)
-
CaF2
1.3×10-3
-
1.6×10-3(298)
1.7×10-3(299)
-
-
-
-
-
-
-
Ca(HCO3)2
16.2
-
16.6
-
17.05
-
17.5
-
17.95
-
18.4
CaI2
64.6
66
67.6
69
70.8
-
74
-
78
-
81
Ca(IO3)2·6H2O
0.09
0.17
0.24
0.38
0.52
-
0.65
-
0.66
0.67
-
Ca(NO2)2·4H2O
63.9
-
84.5(291)
104
-
-
134
-
151
166
178
Ca(NO3)2·4H2O
102
115
129
152
191
-
-
-
358
-
363
Ca(OH)2
0.19
0.182
0.173
0.16
0.141
0.128
0.121
0.106
0.094
0.086
0.076
CaSO4·1/2H2O
-
-
0.32
0.29(298)
0.26(308)
0.21(318)
0.145(338)
0.12(348)
-
-
0.071
CdCl2·2.5H2O
90
100
113
132
-
-
-
-
-
-
-
CdCl2·H2O
-
135
135
135
135
-
136
-
140
-
147
Cl2①
1.46
0.98
0.716
0.562
0.451
0.386
0.324
0.274
0.219
0.125
0
CO①
0
0.0035
0.0028
0.0024
0.002
0.0018
0.0015
0.0013
0.001
6E-04
0
CO2①
0.33
0.2318
0.1688
0.1257
0.097
0.0761
0.0576
-
-
-
0
CoCl2
43.5
47.7
52.9
59.7
69.5
-
93.8
-
97.6
101
106
Co(NO3)2
84
89.6
97.4
111
125
-
174
-
204
300
-
CoSO4
25.5
30.5
36.1
42
48.8
-
55
-
53.8
45.3
38.9
CoSO4·7H2O
44.8
56.3
65.4
73
88.1
-
101
-
-
-
-
CrO3
165
-
167.2
-
172.5
183.9
-
-
191.6
217.5
206.8
CsCl
161
175
187
197
208
218.5
230
239.5
250
260
271
CsOH
-
-
395.5(288)
-
-
-
-
-
-
-
-
CuCl2
68.6
70.9
73
77.3
87.6
-
96.5
-
104
108
120
CuI2
-
-
1.107
-
-
-
-
-
-
-
-
Cu(NO3)2
83.5
100
125
156
163
-
182
-
208
222
247
CuSO4·5H2O
23.1
27.5
32
37.8
44.6
-
61.8
-
83.8
-
114
FeCl2
49.7
59
62.5
66.7
70
-
78.3
-
88.7
92.3
94.9
FeCl3·6H2O
74.4
81.9
91.8
106.8
-
315.1
-
-
525.8
-
535.7
Fe(NO3)2·6H2O
113
134
-
-
-
-
266
-
-
-
-
FeSO4·7H2O
28.8
40
48
60
73.3
-
100.7
-
79.9
68.3
57.8
H3BO3
2.67
3.72
5.04
6.72
8.72
11.54
14.81
18.62
23.62
30.38
40.25
HBr①
221
210.3
204(288)
-
-
171.5
-
-
150.5(348)
-
130
HCl①
82.3
77.2
72.6
67.3
63.3
59.6
56.1
-
-
-
-
H2C2O4
3.54
6.08
9.52
14.23
21.52
-
44.32
-
84.5
125
-
HgBr
-
-
4×10-6(299)
-
-
-
-
-
-
-
-
HgBr2
0.3
0.4
0.56
0.66
0.91
-
1.68
-
2.77
-
4.9
Hg2Cl2
0
-
0.0002
-
7E-04
-
-
-
-
-
-
HgCl2
3.63
4.82
6.57
8.34
10.2
-
16.3
-
30
-
61.3
I2
0.01
0.02
0.029
0.039
0.052
0.078
0.1
-
0.225
0.315
0.445
KBr
53.5
59.5
65.3
70.7
75.4
80.2
85.5
90
95
99.2
104
KBrO3
3.09
4.72
6.91
9.64
13.1
17.5
22.7
-
34.1
-
49.9
KC2H3O2
216
233
256
283
324
-
350
-
381
398
-
K2C2O4
25.5
31.9
36.4
39.9
43.8
-
53.2
-
63.6
69.2
75.3
KCl
28
31.2
34.2
37.2
40.1
42.6
45.8
48.3
51.3
54
56.3
KClO3
3.3
5.2
7.3
10.1
13.9
19.3
23.8
-
37.6
46
56.3
KClO4
0.76
1.06
1.68
2.56
3.73
6.5
7.3
11.8
13.4
17.7
22.3
KSCN
177
198
224
255
289
-
372
-
492
571
675
K2CO3
105
108
111
114
117
121.2
127
133.1
140
148
156
K2CrO4
56.3
60
63.7
66.7
67.8
-
70.1
70.4
72.1
74.5
75.6
K2Cr2O7
4.7
7
12.3
18.1
26.3
34
45.6
52
73
-
80
K3Fe(CN)6
30.2
38
46
53
59.3
-
70
-
-
-
91
K4Fe(CN)6
14.3
21.1
28.2
35.1
41.4
-
54.8
-
66.9
71.5
74.2
KHC4H4O6
0.23
0.358
0.523
0.762
-
-
-
-
-
-
-
KHCO3
22.5
27.4
33.7
39.9
47.5
-
65.6
-
-
-
-
KHSO4
36.2
-
48.6
54.3
61
-
76.4
-
96.1
-
122
KI
128
136
144
153
162
168
176
184
192
198
208
KIO3
4.6
6.27
8.08
10.03
12.6
-
18.3
-
24.8
-
32.3
KMnO4
2.83
4.31
6.34
9.03
12.6
16.98
22.1
-
-
-
-
KNO2
279
292
306
320
329
-
348
-
376
390
410
KNO3
13.9
21.2
31.6
45.3
61.3
85.5
106
138
167
203
245
KOH
95.7
103
112
126
134
140
154
-
-
-
178
K2PtCl6
0.48
0.6
0.78
1
1.36
2.17
2.45
3.19
3.71
4.45
5.03
K2SO4
7.4
9.3
11.1
13
14.8
16.5
18.2
19.75
21.4
22.9
24.1
K2S2O8
1.65
2.67
4.7
7.75
11
-
-
-
-
-
-
K2SO4·Al2(SO4)3
3
3.99
5.9
8.39
11.7
17
24.8
40
71
109
-
LiCl
69.2
74.5
83.5
86.2
89.8
97
98.4
-
112
121
128
Li2CO3
1.54
1.43
1.33
1.26
1.17
1.08
1.01
-
0.85
-
0.72
LiF
-
-
0.27(291)
-
-
-
-
-
-
-
-
LiOH
11.9
12.11
12.35
12.7
13.22
13.3
14.63
-
16.56
-
19.12
Li3PO4
-
-
0.039(291)
-
-
-
-
-
-
-
-
MgBr2
98
99
101
104
106
-
112
-
113.7
-
125
MgCl2
52.9
53.6
54.6
55.8
57.5
-
61
-
66.1
69.5
73.3
MgI2
120
-
140
-
173
-
-
-
186
-
-
Mg(NO3)2
62.1
66
69.5
73.6
78.9
-
78.9
-
91.6
106
-
Mg(OH)2
-
-
0.0009(291)
-
-
-
-
-
-
-
0.004
MgSO4
22
28.2
33.7
38.9
44.5
-
54.6
-
55.8
52.9
50.4
MnCl2
63.4
68.1
73.9
80.8
88.5
98.15
109
-
113
114
115
Mn(NO3)2
102
118
139
206
-
-
-
-
-
-
-
MnC2O4
0.02
0.024
0.028
0.033
-
-
-
-
-
-
-
MnSO4
52.9
59.7
62.9
62.9
60
-
53.6
-
45.6
40.9
35.3
NH4Br
60.5
68.1
76.4
83.2
91.2
99.2
108
116.8
125
135
145
NH4SCN
120
144
170
208
234
-
346
-
-
-
-
(NH4)2C2O4
2.2
3.21
4.45
6.09
8.18
10.3
14
-
22.4
27.9
34.7
NH4Cl
29.4
33.3
37.2
41.4
45.8
50.4
55.3
60.2
65.6
71.2
77.3
NH4ClO4
12
16.4
21.7
27.7
34.6
-
49.9
-
68.9
-
-
(NH4)2·Co(SO4)2
6
9.5
13
17
22
27
33.5
40
49
58
75.1
(NH4)2CrO4
25
29.2
34
39.3
45.3
-
59
-
76.1
-
-
(NH4)2Cr2O7
18.2
25.5
35.6
46.5
58.5
-
86
-
115
-
156
(NH4)2·Cr2(SO4)4
3.95
-
10.78(298)
18.8
32.6
-
-
-
-
-
-
(NH4)2·Fe(SO4)2
12.5
17.2
-
-
33
40
-
52
-
-
-
(NH4)2·Fe2(SO4)4
-
-
-
44.15(298)
-
-
-
-
-
-
-
NH4HCO3
11.9
16.1
21.7
28.4
36.6
-
59.2
-
109
170
354
NH4H2PO4
22.7
29.5
37.4
46.4
56.7
-
82.5
-
118
-
173
(NH4)2HPO4
42.9
62.9
68.9
75.1
81.8
-
97.2
-
-
-
-
NH4I
155
163
172
182
191
199.6
209
218.7
229
-
250
NH4MgPO4
0.02
-
0.052
-
0.036
0.03
0.04
0.016
0.019
-
0.0195
NH4MnPO4·H2O
-
0.0031(冷水)
-
-
-
-
-
0.05(热水)
-
-
-
NH4NO3
118
-
192
241.8
297
344
421
499
580
740
871
(NH4)2PtCl6
0.29
0.374
0.499
0.637
0.815
-
1.44
-
2.16
2.61
3.36
(NH4)2SO4
70.6
73
75.4
78
81
-
88
-
95
-
103
(NH4)2SO4·Al2(SO4)3
2.1
5
7.74
10.9
14.9
20.1
26.7
-
-
-
109.7(368)
(NH4)2S2O8
58.2
-
-
-
-
-
-
-
-
-
-
(NH4)3SbS4
71.2
-
91.2
120
-
-
-
-
-
-
-
(NH4)2SeO4
-
117(280)
-
-
-
-
-
-
-
-
197
NH4VO3
-
-
0.48
0.84
1.32
1.78
2.42
3.05
-
-
-
NaBr
80.2
85.2
90.8
98.4
107
116
118
-
120
121
121
Na2B4O7
1.11
1.6
2.56
3.86
6.67
10.5
19
24.4
31.4
41
52.5
NaBrO3
24.2
30.3
36.4
42.6
48.8
-
62.6
-
75.7
-
90.8
NaC2H3O2
36.2
40.8
46.4
54.6
65.6
83
139
146
153
161
170
Na2C2O4
2.69
3.05
3.41
3.81
4.18
-
4.93
-
5.71
-
6.5
NaCl
35.7
35.8
35.9
36.1
36.4
37
37.1
37.8
38
38.5
39.2
NaClO3
79.6
87.6
95.9
105
115
-
137
-
167
184
204
Na2CO3
7
12.5
21.5
39.7
49
-
46
-
43.9
43.9
-
Na2CrO4
31.7
50.1
84
88
96
104
115
123
125
-
126
Na2Cr2O7
163
172
183
198
215
244.8
269
316.7
376
405
415
Na4Fe(CN)6
11.2
14.8
18.8
23.8
29.9
-
43.7
-
62.1
-
-
NaHCO3
7
8.1
9.6
11.1
12.7
14.45
16
-
-
-
-
NaH2PO4
56.5
69.8
86.9
107
133
157
172
190.3
211
234
-
Na2HPO4
1.68
3.53
7.83
22
55.3
80.2
82.8
88.1
92.3
102
104
NaI
159
167
178
191
205
227.8
257
294
295
-
302
NaIO3
2.48
2.59
8.08
10.7
13.3
-
19.8
-
26.6
29.5
33
NaNO3
73
80.8
87.6
94.9
102
104.1
122
-
148
-
180
NaNO2
71.2
75.1
80.8
87.6
94.9
-
111
-
133
-
160
NaOH
-
98
109
119
129
-
174
-
-
-
-
Na3PO4
4.5
8.2
12.1
16.3
20.2
-
29.9
-
60
68.1
77
Na4P2O7
3.16
3.95
6.23
9.95
13.5
17.45
21.83
-
30.04
-
40.2
- 配套讲稿:
如PPT文件的首页显示word图标,表示该PPT已包含配套word讲稿。双击word图标可打开word文档。
- 特殊限制:
部分文档作品中含有的国旗、国徽等图片,仅作为作品整体效果示例展示,禁止商用。设计者仅对作品中独创性部分享有著作权。
- 关 键 词:
- 常见 物质 溶解度
 冰豆网所有资源均是用户自行上传分享,仅供网友学习交流,未经上传用户书面授权,请勿作他用。
冰豆网所有资源均是用户自行上传分享,仅供网友学习交流,未经上传用户书面授权,请勿作他用。


 铝散热器项目年度预算报告.docx
铝散热器项目年度预算报告.docx
