 FDA溶出度方法数据库05.docx
FDA溶出度方法数据库05.docx
- 文档编号:9969440
- 上传时间:2023-02-07
- 格式:DOCX
- 页数:201
- 大小:57.82KB
FDA溶出度方法数据库05.docx
《FDA溶出度方法数据库05.docx》由会员分享,可在线阅读,更多相关《FDA溶出度方法数据库05.docx(201页珍藏版)》请在冰豆网上搜索。
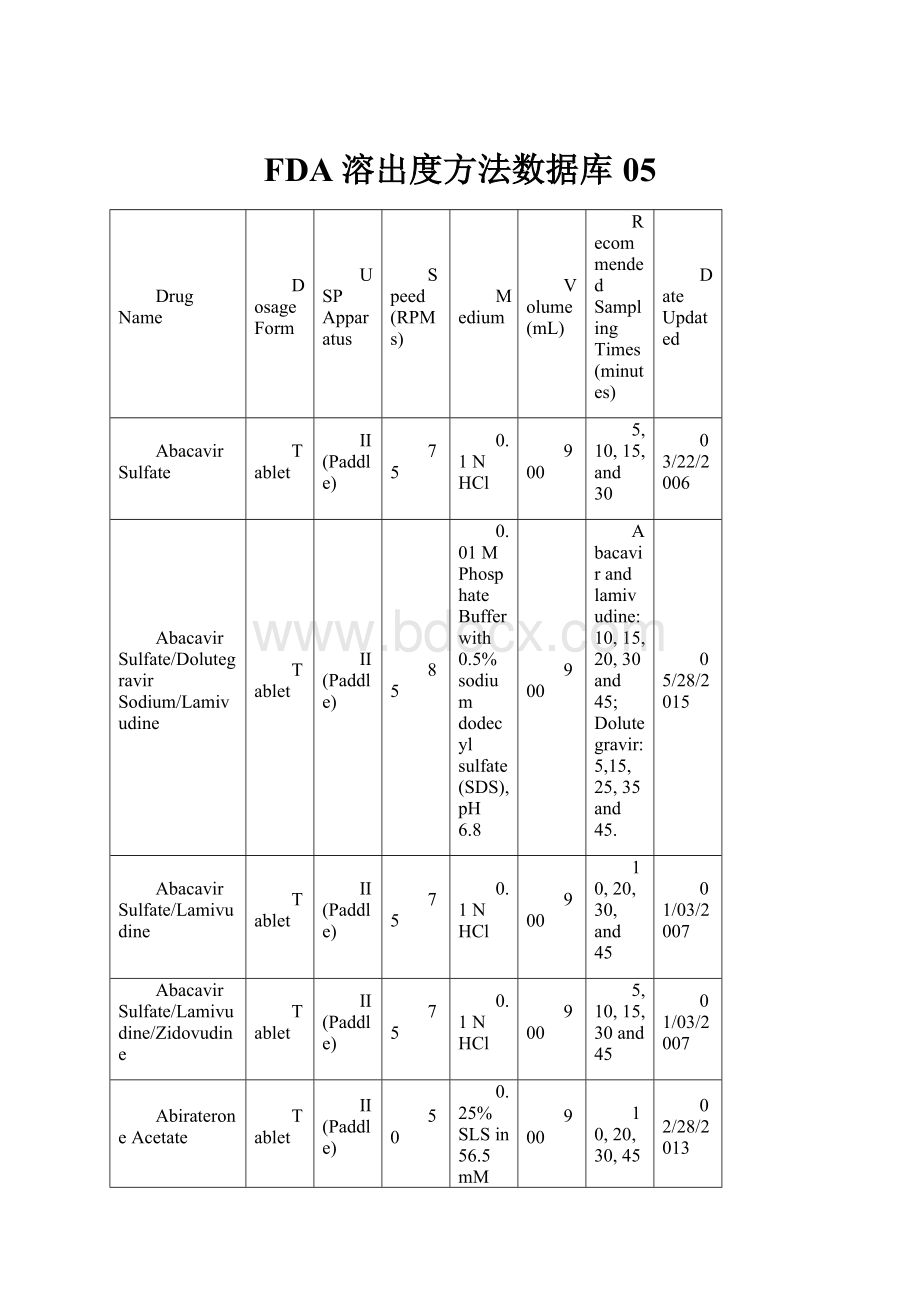
FDA溶出度方法数据库05
DrugName
DosageForm
USP
Apparatus
Speed(RPMs)
Medium
Volume(mL)
RecommendedSamplingTimes(minutes)
DateUpdated
AbacavirSulfate
Tablet
II(Paddle)
75
0.1NHCl
900
5,10,15,and30
03/22/2006
AbacavirSulfate/DolutegravirSodium/Lamivudine
Tablet
II(Paddle)
85
0.01MPhosphateBufferwith0.5%sodiumdodecylsulfate(SDS),pH6.8
900
Abacavirandlamivudine:
10,15,20,30and45;Dolutegravir:
5,15,25,35and45.
05/28/2015
AbacavirSulfate/Lamivudine
Tablet
II(Paddle)
75
0.1NHCl
900
10,20,30,and45
01/03/2007
AbacavirSulfate/Lamivudine/Zidovudine
Tablet
II(Paddle)
75
0.1NHCl
900
5,10,15,30and45
01/03/2007
AbirateroneAcetate
Tablet
II(Paddle)
50
0.25%SLSin56.5mMphosphatebuffer,pH4.5
900
10,20,30,45and60
02/28/2013
AcamprosateCalcium
Tablet(DelayedRelease)
I(Basket)
180
AcidStage:
0.1NHClBufferStage:
"Citrate-sodiumhydroxide"bufferpH6.8(150mlof2NNaOH,21.014gmofcitricacidandultra-purewaterto1000ml)(MethodB)
1000
120(Acid)30,60,90,120,and180(buffer)
12/20/2005
Acarbose
Tablet
II(Paddle)
75
Water(deaerated)
900
10,15,20,30and45
03/22/2006
Acetaminophen
Suppository
II(Paddle)
50
Phosphatebuffer,pH5
900
15,30,45,60and90
08/17/2006
Acetaminophen
Tablet(ExtendedRelease)
RefertoUSP
03/03/2011
Acetaminophen/Butalbital
Tablet
II(Paddle)
50
Water(deaerated)
900
15,30,45,60and90
01/03/2007
Acetaminophen/Butalbital/Caffeine
Tablet
RefertoUSP
01/14/2008
Acetaminophen/Butalbital/Caffeine/CodeinePhosphate
Capsule
II(Paddle)
50
Water(deaerated)
1000
10,20,30,45and60
05/15/2014
Acetaminophen/Caffeine/DihydrocodeineBitartrate
Tablet
II(Paddle)
50
Water
900
10,15,30,45and60
07/25/2007
Acetaminophen/Caffeine/DihydrocodeineBitartrate
Capsule
I(Basket)
100
Water
900
10,20,30,45and60
01/03/2007
Acetaminophen/HydrocodoneBitartrate
Tablet
RefertoUSP(provideindividualunitdata).
Acetaminophen/Oxycodone
Tablet
RefertoUSP
01/14/2008
Acetaminophen/PentazocineHCl
Tablet
I(Basket)
100
Water(deaerated)
900
10,20,30,45and60
01/12/2004
Acetaminophen/PropoxypheneHCl
Tablet
RefertoUSP
01/15/2010
Acetaminophen/PropoxypheneNapsylate
Tablet
RefertoUSP
01/15/2010
Acetaminophen/TramadolHCl
Tablet
II(Paddle)
50
0.1NHCl
900
5,10,15,20and30
03/04/2006
Acetazolamide
Capsule(ExtendedRelease)
II(Paddle)
75
AcetateBuffer,pH4.5with2.2%Tween20
900
1,2,5,7,9,12and14hours
01/15/2010
Acetazolamide
Tablet
RefertoUSP
07/21/2011
Acetazolomide
Tablet
RefertoUSP
07/14/2008
Acitretin
Capsule
RefertoUSP
09/22/2011
Acrivastine/PseudoephedrineHCl
Capsule
II(Paddle)
50
0.01NHCl
900
5,10,15and30
01/12/2004
Acyclovir
Suspension
II(Paddle)
50
0.1NHCl
900
10,20,30,45and60
02/20/2004
Acyclovir
Capsule
RefertoUSP
01/05/2012
Acyclovir
Tablet
RefertoUSP
06/18/2007
AdefovirDipivoxil
Tablet
II(Paddle)
50
0.01NHCl
600
10,20,30,45and60
04/10/2008
AfatinibDimaleate
Tablet
II(Paddle)
75
McIlvaineBufferpH4.0
900
5,10,15,20and30
05/28/2015
Albendazole
Tablet
RefertoUSP
08/15/2013
AlbuterolSulfate
Tablet(ExtendedRelease)
II(Paddle)
50
0.1NHCl
900
1,2,4,6,9and12hours
04/09/2007
AlbuterolSulfate
Tablet
RefertoUSP
09/03/2008
AlendronateSodium
Tablet
RefertoUSP
01/14/2008
AlendronateSodium/Cholecalciferol
Tablet
II(Paddle)
ForAlendronate:
50;ForCholecalciferol:
75
ForAlendronate:
DeaeratedWater;ForCholecalciferol:
0.3%SDSinUSPWater
ForAlendronate:
900;ForCholecalciferol:
500
10,15,20,30and45
11/25/2008
AlfuzosinHCl
Tablet(ExtendedRelease)
II(Paddle)
100
0.01NHCl
900
1,2,12,20hours
06/18/2007
AliskirenHemifumarate
Tablet
I(Basket)
100
0.01NHCl
500
10,20,30and45
09/03/2008
AliskirenHemifumarate/AmlodipineBesylate
Tablet
I(Basket)
100
0.01NHCl,pH2.0
500
10,15,20,30and45
06/07/2012
AliskirenHemifumarate/AmlodipineBesylate/Hydrochlorothiazide
Tablet
I(Basket)
100
0.01NHCl
900
10,15,20,30and45
06/07/2012
AliskirenHemifumarate/Hydrochlorothiazide
Tablet
I(Basket)
100
0.1NHCl
900
10,15,20,30and45
10/08/2009
AliskirenHemifumarate/Valsartan
Tablet
I(Basket)
100
PhosphateBuffer,pH6.8
1000
5,10,15,20,30and45
12/23/2010
Allopurinol
Tablet
RefertoUSP
07/25/2007
AlmotriptanMalate
Tablet
II(Paddle)
50
0.1NHCl
900
5,10,15,and30
01/20/2006
AlogliptinBenzoate
Tablet
II(Paddle)
50
0.01NHCl
900
5,10,15,20and30
02/14/2014
AlosetronHCl
Tablet
II(Paddle)
50(for1mg)&75(for0.5mg)
Water(deaerated)
500
10,20,30and45
01/26/2006
Alprazolam
Tablet
RefertoUSP
06/18/2007
Alprazolam
Tablet(OrallyDisintegrating)
II(Paddle)
50
70mMPotassiumPhosphateBuffer,pH6.0
500
2,5,10,15and20
10/06/2008
Alprazolam
Tablet(ExtendedRelease)
I(Basket)
100
1%PhosphateBuffer,pH6.0
500
1,4,8,12and16hours
02/08/2007
Altretamine
Capsule
RefertoUSP
01/29/2010
Alvimopan
Capsule
II(Paddle
50
0.1NHCl
900
5,10,15,20,30and45
10/21/2010
AmantadineHCl
Tablet
II(Paddle)
50
Water(deaerated)
500
10,20,30,45and60
01/12/2004
AmantadineHCl
Capsule
RefertoUSP
12/23/2010
Ambrisentan
Tablet
II(Paddle)
75
0.05MAcetateBuffer,pH5.0
900
5,10,15,30,and45
05/20/2009
AmilorideHCl
Tablet
RefertoUSP
06/07/2012
AmilorideHCl/Hydrochlorothiazide
Tablet
RefertoUSP
06/07/2012
Aminosalicylic
Granule(DelayedRelease)
II(Paddle)
100
AcidStage:
0.1NHCl;BufferStage2:
pH7.5PhosphateBuffer
1000
AcidStage:
2hours;BufferStage:
1,2,3and4hours
07/14/2008
AmiodaroneHCl(Test1)
Tablet
II(Paddle)
100
1%SLSinwater
1000
10,20,30,45,60and90
01/12/2004
AmiodaroneHCl(Test2)
Tablet
I(Basket)
50
AcetateBuffer,pH4.0,with1%Tween80
900
10,20,30,45,60and90
01/12/2004
AmitriptylineHCl
Tablet
RefertoUSP
01/14/2008
AmlodipineBesylate
Tablet(OrallyDisintegrating)
II(Paddle)
50
0.01NHCl
500
5,10,15and20
10/06/2008
AmlodipineBesylate
Tablet
II(Paddle)
75
0.01NHCl
500
10,20,30,45and60
01/14/2004
AmlodipineBesylate/AtorvastatinCalcium
Tablet
II(Paddle)
75
PhosphateBuffer,pH6.8
900
5,10,15and30
04/02/2009
AmlodipineBesylate/BenazeprilHCl
Capsule
I(Basket)
100
0.01NHCl
500
10,20,30,45,and60
06/20/2007
AmlodipineBesylate/Hydrochlorothiazide/OlmesartanMedoxomil
Tablet
II(Paddle)
50
PhosphateBuffer,pH6.8
900
5,10,15,20,30and45
07/21/2011
AmlodipineBesylate/Hydrochlorothiazide/Valsartan
Tablet
II(Paddle)
50
PhosphateBuffer,pH6.8
900
10,20,30and45
03/25/2010
AmlodipineBesylate/Hydrochlorothiazide/Valsartan(10/25/320mg)
Tablet
II(Paddle)
55
PhosphateBuffer,pH6.8
900
10,20,30and45
03/25/2010
AmlodipineBesylate/OlmesartanMedoxomil
Tablet
II(Paddle)
50
PhosphateBuffer,pH6.8
900
10,20,30and45
08/11/2008
AmlodipineBesylate/Telmisartan
Tablet
II(Paddle)
75
Telmisartan:
PhosphateBuffer,pH7.5;Amlodipine:
0.01NHCl,pH2
Telmisartan:
900;Amlodipine:
500
Telmisartan:
10,15,20,30and45;Amlodipine:
10,15,20,30and45
08/05/2010
AmlodipineBesylate/Valsartan
Tablet
II(Paddle)
75
PhosphateBuffer,pH6.8
1000
5,10,15,20,30and45
07/21/2011
Amoxicillin
Tablet(ExtendedRelease)
II(Paddle)
100
3Stagedissolution:
50mMpotassiumphosphatemonobasicbufferatpH4.0(0-2hours),6.0(2-4hours)and6.8(4hoursandbeyond)
900
0.25,0.5,1,2,2.25,2.5,3,4,4.25,4.5,5and6hours
10/21/2010
Amoxicillin
Capsule
RefertoUSP
01/31/2013
Amoxicillin
Tablet
RefertoUSP
01/31/2013
Amoxicillin
ForOralSuspension
II(Paddle)
50
Water(degassed)
900
5,10,15,20,30and45
06/06/2013
Amoxicillin/Clarithromycin/Lansoprazole
Capsule/Tablet/Capsule(Copackage)
RefertoUSPformonographsofAmoxicillinCapsules,ClarithromycinTabletsandLansoprazoleDelayed-ReleaseCapsules
02/28/2013
Amoxicillin/Clarithromycin/Omeprazole
Capsule/Tablet/Capsule(Copackage)
RefertoUSPformonographsofAmoxicillinCapsules,ClarithromycinTabletsandOmeprazoleDelayed-ReleaseCapsules
02/28/2013
Amoxicillin/ClavulanatePotassium
Tablet
RefertoUSP
Asappropriate
0,0.5,1,2,3,4and5hours
10/04/2012
Amoxicillin/ClavulanatePotassium
Suspension
II(Paddle)
75
Water(deaerated)
900
5,10,15and30
01/14/2004
Amoxicillin/ClavulanatePotassium
Tablet(Chewable)
RefertoUSP
01/14/2008
AmphetamineAspartate/AmphetamineSulfate/DextroamphetamineSaccharate/DextroamphetamineSulfate
Capsule(ExtendedRelease)
II(Paddle)
50
DiluteHCl,pH1.1forfirst2hrs,thenadd200mLof200mMPhosphateBufferandadjusttopH6.0fortheremainder
0-2hrs:
750mL.After2hrs:
950mL
0.5,1,2,3,and4hours
07/25/2007
AmphetamineAspartate/AmphetamineSulfate/De
- 配套讲稿:
如PPT文件的首页显示word图标,表示该PPT已包含配套word讲稿。双击word图标可打开word文档。
- 特殊限制:
部分文档作品中含有的国旗、国徽等图片,仅作为作品整体效果示例展示,禁止商用。设计者仅对作品中独创性部分享有著作权。
- 关 键 词:
- FDA 溶出度 方法 数据库 05
 冰豆网所有资源均是用户自行上传分享,仅供网友学习交流,未经上传用户书面授权,请勿作他用。
冰豆网所有资源均是用户自行上传分享,仅供网友学习交流,未经上传用户书面授权,请勿作他用。


 1212中级汽车维修工考试试题三.docx
1212中级汽车维修工考试试题三.docx
