 Atorvastatin Calcium USP37 Word version.docx
Atorvastatin Calcium USP37 Word version.docx
- 文档编号:9914493
- 上传时间:2023-02-07
- 格式:DOCX
- 页数:20
- 大小:45.64KB
Atorvastatin Calcium USP37 Word version.docx
《Atorvastatin Calcium USP37 Word version.docx》由会员分享,可在线阅读,更多相关《Atorvastatin Calcium USP37 Word version.docx(20页珍藏版)》请在冰豆网上搜索。
AtorvastatinCalciumUSP37Wordversion
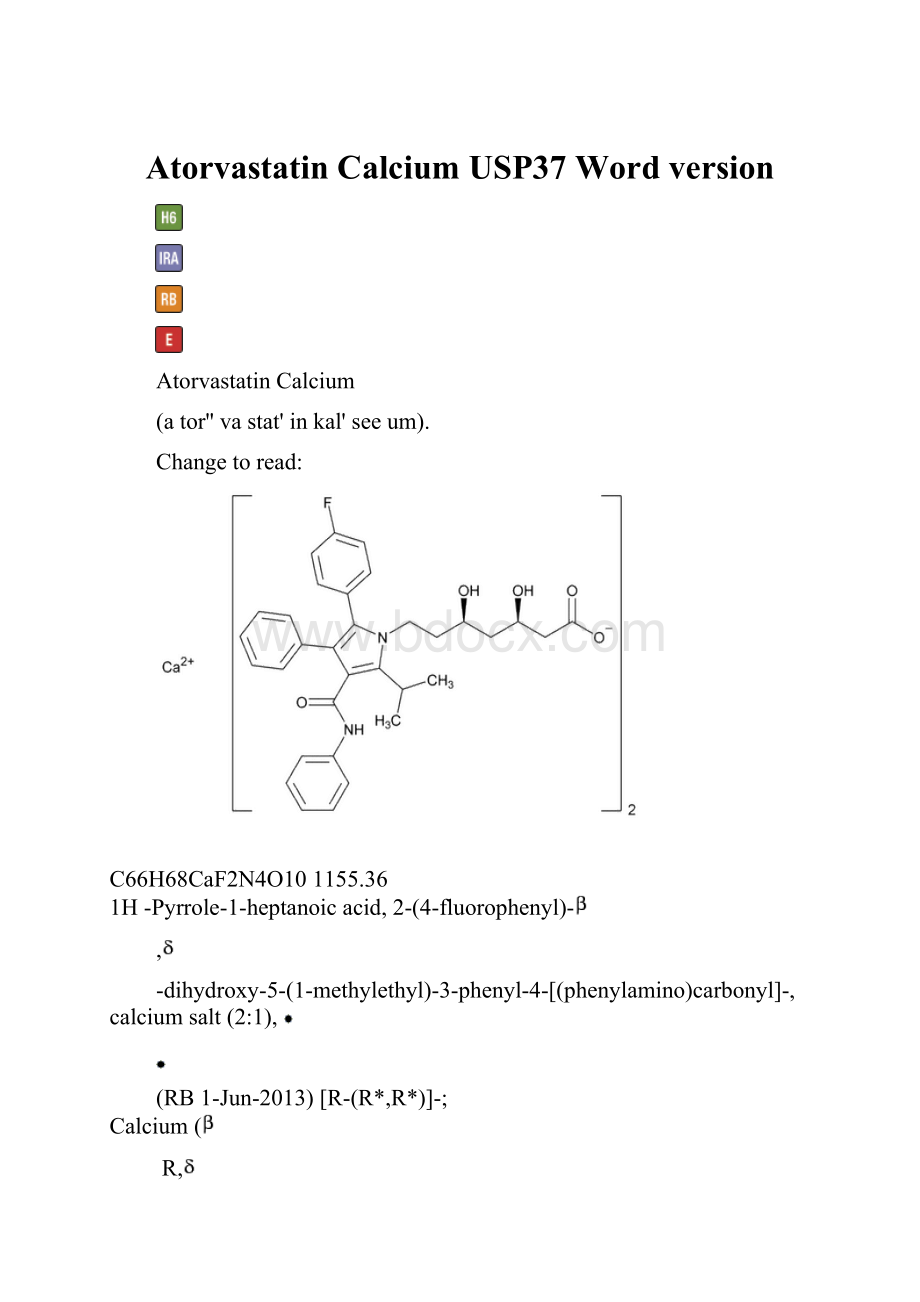
AtorvastatinCalcium
(ator''vastat'inkal'seeum).
Changetoread:
C66H68CaF2N4O101155.36
1H-Pyrrole-1-heptanoicacid,2-(4-fluorophenyl)-
-dihydroxy-5-(1-methylethyl)-3-phenyl-4-[(phenylamino)carbonyl]-,calciumsalt(2:
1),
(RB1-Jun-2013)[R-(R*,R*)]-;
Calcium(
R,
R)-2-(p-fluorophenyl)-
-dihydroxy-5-isopropyl-3-phenyl-4-(phenylcarbamoyl)pyrrole-1-heptanoate(1:
2);
[(3R,5R)-7-[3-(Phenylcarbamoyl)-5-(4-fluorophenyl)-2-isopropyl-4-phenyl-1H-pyrrol-1-yl]-3,5-dihydroxyheptanoicacid,calciumsalt]
(RB1-Jun-2013)
Anhydrous
[134523-03-8].
C66H68CaF2N4O10·3H2O1209.41
Trihydrate
[344423-98-9].
C66H68CaF2N4O10·C3H8O2
Propyleneglycolsolvate 1231.46
(RB1-Jun-2013)
DEFINITION
Changetoread:
AtorvastatinCalciumcontainsNLT98.0%andNMT102.0%ofatorvastatincalcium(C66H68CaF2N4O10),calculatedontheanhydrous
andsolvent-freebasis.Iflabeledasapropyleneglycolsolvate,itcontainsNLT98.0%andNMT102.0%ofatorvastatincalcium(C66H68CaF2N4O10),calculatedontheanhydrous,propyleneglycol-free,andsolvent-freebasis.Itmaycontainasuitableantioxidant.
(RB1-Jun-2013)
IDENTIFICATION
Changetoread:
• A.InfraredAbsorption
197K
:
[
Note—IfadifferenceappearsintheIRspectraoftheanalyteandthestandard,separatelydissolveequalportionsofthesamplespecimenandtheUSPReferenceStandardinequalvolumesofmethanol,evaporatethesolutiontodrynessinsimilarcontainersunderidenticalconditions,andrepeatthetestontheresidues.]
(RB1-Jun-2013)
• B.Calcium
Diluent:
Methanol,water,andhydrochloricacid(75:
25:
2)
Samplesolution:
0.05mg/mLofAtorvastatinCalciuminDiluent
Blank:
Diluent
Analysis
Samples:
SamplesolutionandBlank
Instrumentalconditions
(SeeSpectrophotometryandLight-Scattering
851
.)
Mode:
Atomicabsorptionspectrophotometry
Analyticalwavelength:
Calciumemissionlineat422.7nm
Flame:
Air–acetylene
Acceptancecriteria:
TheSamplesolutionexhibitsasignificantabsorptionatthecalciumemissionlineat422.7nm.
ASSAY
Changetoread:
• Procedure
Buffer:
3.9g/Lofammoniumacetateinwater.AdjustwithglacialaceticacidtoapHof5.0±0.1.
SolutionA:
Acetonitrile,stabilizer-freetetrahydrofuran,andBuffer(21:
12:
67)
SolutionB:
Acetonitrile,stabilizer-freetetrahydrofuran,andBuffer(61:
12:
27)
Mobilephase:
SeeTable1.[Note—Ifnecessary,adjusttheMobilephasebyincreasingordecreasingthepercentageofacetonitrileorthepHoftheammoniumacetatesolutiontoachievearetentiontimeof26–34minfortheatorvastatinpeak.]
Table1
Time
(min)
SolutionA
(%)
SolutionB
(%)
0
100
0
40
100
0
70
20
80
85
0
100
100
0
100
105
100
0
115
100
0
Diluent:
N,N-dimethylformamide
Systemsuitabilitysolution:
0.05mg/mLofUSPAtorvastatinCalciumRSand0.06mg/mLofUSPAtorvastatinRelatedCompoundBRSinDiluent
Standardsolution:
0.4mg/mLofUSPAtorvastatinCalciumRSinDiluent.[Note—Usesonicationifnecessary.]
Samplesolution:
0.4mg/mLofAtorvastatinCalciuminDiluent.[Note—Usesonicationifnecessary.]
Chromatographicsystem
(SeeChromatography
621
SystemSuitability.)
[Note—IfsignificantfrontingofthepeaksforatorvastatinrelatedcompoundBandatorvastatinisobserved,usethefollowingdiluenttopreparetheSamplesolution,theStandardsolution,andtheSystemsuitabilitysolution:
acetonitrile,stabilizer-freetetrahydrofuran,andwater(1:
1:
2).]
Mode:
LC
Detector:
UV244nm
Column:
4.6-mm×25-cm;5-µmpackingL7
Columntemperature:
35
Flowrate:
1.5mL/min
Injectionvolume:
20µL
Systemsuitability
Samples:
SystemsuitabilitysolutionandStandardsolution
Suitabilityrequirements
Resolution:
NLT1.5betweenthepeaksforatorvastatinrelatedcompoundBandatorvastatin,Systemsuitabilitysolution
Tailingfactor:
NMT1.6,Standardsolution
Relativestandarddeviation:
NMT0.6%,Standardsolution
Analysis
Samples:
StandardsolutionandSamplesolution
Calculatethepercentageofatorvastatincalcium(C66H68CaF2N4O10)intheportionofAtorvastatinCalciumtaken:
Result=(rU/rS)×(CS/CU)×100
rU
=
=peakresponsefromtheSamplesolution
rS
=
=peakresponsefromtheStandardsolution
CS
=
=concentrationofUSPAtorvastatinCalciumRSintheStandardsolution(mg/mL)
CU
=
=concentrationofAtorvastatinCalciumintheSamplesolution(mg/mL)
Acceptancecriteria:
98.0%–102.0%ontheanhydrous
andsolvent-freebasis.Iflabeledasapropyleneglycolsolvate,98.0%–102.0%ontheanhydrous,propyleneglycol-free,andsolvent-freebasis.
(RB1-Jun-2013)
OTHERCOMPONENTS
Addthefollowing:
•
ContentofPropyleneGlycol(iflabeledasapropyleneglycolsolvate)
Diluent:
Dimethylsulfoxide
Standardsolution:
0.125mg/mLofpropyleneglycolinDiluent
Samplesolution:
2.5mg/mLofAtorvastatinCalcium(aspropyleneglycolsolvate)inDiluent.Usesonicationasneededtoachieveacompletedissolution.
Chromatographicsystem
(SeeChromatography
621
SystemSuitability.)
Mode:
GC
Detector:
Flameionization
Column:
0.53-mm×75-m;3-µmcoatingofG43
Temperatures
Injectionport:
230
Detector:
250
Column:
SeeTable2.
Table2
Initial
Temperature
(
)
Temperature
Ramp
(
/min)
Final
Temperature
(
)
HoldTimeatFinal
Temperature
(min)
100
0
100
1
100
10
140
5
140
30
225
3
Carriergas:
Helium
Flowrate:
6.0mL/min
Injectionvolume:
1µL
Injectiontype:
Splitless,usingasuitableinletliner
Systemsuitability
Sample:
Standardsolution
Suitabilityrequirements
Tailingfactor:
NMT2.0
Relativestandarddeviation:
NMT5.0%
Analysis
Samples:
StandardsolutionandSamplesolution
CalculatethepercentageofpropyleneglycolintheportionofAtorvastatinCalciumaspropyleneglycolsolvatetaken:
Result=(rU/rS)×(CS/CU)×100
rU
=
=peakresponseofpropyleneglycolfromtheSamplesolution
rS
=
=peakresponseofpropyleneglycolfromtheStandardsolution
CS
=
=concentrationofpropyleneglycolintheStandardsolution(mg/mL)
CU
=
=concentrationofAtorvastatinCalcium(aspropyleneglycolsolvate)intheSamplesolution(mg/mL)
Acceptancecriteria:
5.4%–7.3%
(RB1-Jun-2013)
IMPURITIES
• HeavyMetals
Diluent:
Methanolandwater(9:
1)
Samplesolution:
Dissolve250mgofthesamplein30mLofDiluent.
Standardleadsolution:
PrepareasdirectedinHeavyMetals
231
.
Referencesolution:
Dilute0.5mLoftheStandardleadsolutionwithDiluentto30mL.
Blanksolution:
20mLofDiluent
Monitorsolution:
Dissolve250mgofAtorvastatinCalciumin0.5mLoftheStandardleadsolution,anddilutewithDiluentto30mL.
Analysis
Samples:
Samplesolution,Referencesolution,Blanksolution,andMonitorsolution
Toeachsolution,add2mLofpH3.5AcetateBufferpreparedasdirectedinHeavyMetals
231
.Mix,addto1.2mLofthioacetamide–glycerinbaseTS,andmiximmediately.Passthesolutionsthroughamembranefilterof0.45-µmporesize.Comparethespotsonthefiltersobtainedwiththedifferentsolutions.ThebrowncolorofthespotfromtheSamplesolutionisnotmoreintensethanthatofthespotfromtheReferencesolution.ThetestisinvalidiftheReferencesolutiondoesnotshowaslightbrowncolorcomparedtotheBlanksolution,orifthecoloroftheMonitorsolutionisnotatleastasintenseasthecoloroftheReferencesolution.
Acceptancecriteria:
NMT20ppm
Changetoread:
• OrganicImpurities,
Procedure1:
[Note—Onthebasisofthesyntheticrouteorofthesolidstatenatureofthedrugsubstance,performeitherProcedure1orProcedure2.Procedure2maybesuitablewhenatorvastatinlactone,atorvastatinepoxytetrahydrofurananalog,andatorvastatinacetonidearepossiblerelatedcompounds,anditmaybesuitableforanamorphousformofthedrugsubstance.]
(RB1-Jun-2013)
Buffer,SolutionA,SolutionB,Mobilephase,Diluent,Systemsuitabilitysolution,andChromatographicsystem:
ProceedasdirectedintheAssay.
Standardsolution:
1.5µg/mLeachofUSPAtorvastatinRelatedCompoundARS,USPAtorvastatinRelatedCompoundBRS,USPAtorvastatinRelatedCompoundCRS,andUSPAtorvastatinRelatedCompoundDRSinDiluent
Samplesolution:
1mg/mLofAtorvastatinCalciuminDiluent.[Note—Usesonicationifnecessary.]
Systemsuitability
Sample:
Systemsuitabilitysolution
Suitabilityrequirements
Resolution:
NLT1.5betweenthepeaksforatorvastatinrelatedcompoundBandatorvastatin
(RB1-Jun-2013)
Analysis
Samples:
StandardsolutionandSamplesolution
ChromatographtheStandardsolution,andidentifythecomponentsbasedontheirrelativeretentiontimes,giveninTable3.
CalculatethepercentageofeachoftheatorvastatinrelatedcompoundsA,B,C,andDintheportionofAtorvastatinCalciumtaken:
Result=(rU/rS)×(CS/CU)×100
rU
=
=peakresponseoftherelevantatorvastatinrelatedcompoundfromtheSamplesolution
rS
=
=peakresponseoftherelevantatorvastatinrelatedcompoundfromtheStandardsolution
CS
=
=concentrationoftherelevantatorvastatinrelatedcompoundintheStandardsolution(mg/mL)
CU
=
=concentrationofAtorvastatinCalciumintheSamplesolution(mg/mL)
CalculatethepercentageofanyotherindividualimpurityintheportionofAtorvastatinCalciumtaken:
Result=(rU/rT)×100
rU
=
=peakresponseofanyotherindividualimp
- 配套讲稿:
如PPT文件的首页显示word图标,表示该PPT已包含配套word讲稿。双击word图标可打开word文档。
- 特殊限制:
部分文档作品中含有的国旗、国徽等图片,仅作为作品整体效果示例展示,禁止商用。设计者仅对作品中独创性部分享有著作权。
- 关 键 词:
- Atorvastatin Calcium USP37 Word version
 冰豆网所有资源均是用户自行上传分享,仅供网友学习交流,未经上传用户书面授权,请勿作他用。
冰豆网所有资源均是用户自行上传分享,仅供网友学习交流,未经上传用户书面授权,请勿作他用。


 《城市规划基本知识》深刻复习要点.docx
《城市规划基本知识》深刻复习要点.docx
